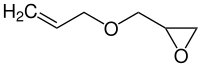
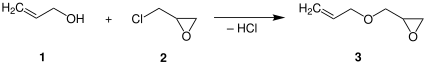烯丙基縮水甘油醚
外觀
此條目可參照英語維基百科相應條目來擴充。 (2024年9月12日) |
| 烯丙基縮水甘油醚 | |
|---|---|
| |
| IUPAC名 2-(prop-2-enoxymethyl)oxirane | |
| 別名 | 2-[(Allyloxy)methyl]oxirane 1-Allyloxy-2,3-epoxypropane Glycidyl allyl ether [(2-Propenyloxy)methyl] oxirane[1] |
| 識別 | |
| CAS號 | 106-92-3 |
| PubChem | 7838 |
| ChemSpider | 13836520 |
| SMILES |
|
| InChI |
|
| InChIKey | LSWYGACWGAICNM-UHFFFAOYAR |
| EINECS | 203-442-4 |
| 性質 | |
| 化學式 | C6H10O2 |
| 摩爾質量 | 114.14 g·mol−1 |
| 外觀 | 無色液體[1] |
| 氣味 | pleasant[1] |
| 密度 | 0.97 g/mL (20 °C)[1] |
| 熔點 | -100 °C(173 K) |
| 沸點 | 154 °C(427 K) |
| 溶解性(水) | 14% (20°C)[1] |
| 溶解性(有機溶劑) | 混溶(丙酮、甲苯、辛烷)[2] |
| 蒸氣壓 | 2 mmHg (20 °C)[1] |
| 折光度n D |
1.4348 (20 °C)[2][3] |
| 危險性 | |
| GHS提示詞 | 危險 |
| H-術語 | H226, H351, H341, H332, H302, H335, H315, H318, H317, H412 |
| 主要危害 | poisonous, mild irritant[2] |
| PEL | 10 ppm (45 mg/m3)[1] |
| 致死量或濃度: | |
LC50(中位濃度)
|
270 ppm (mouse, 4 hr) 670 ppm (rat, 8 hr)[4] |
| 若非註明,所有數據均出自標準狀態(25 ℃,100 kPa)下。 | |
烯丙基縮水甘油醚是一種有機化合物,化學式為C6H10O2。
合成
[編輯]烯丙基縮水甘油醚在商業上通過環氧氯丙烷和烯丙醇在鹼存在下醚化製得:[5]
參考文獻
[編輯]- ^ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 NIOSH Pocket Guide to Chemical Hazards. #0019. NIOSH.
- ^ 2.0 2.1 2.2 CID 7838 from PubChem
- ^ Clayton, G. D. and F. E. Clayton (eds.). Patty's Industrial Hygiene and Toxicology: Volume 2A, 2B, 2C: Toxicology. 3rd ed. New York: John Wiley Sons, 1981-1982., p. 2199
- ^ Allyl glycidyl ether. Immediately Dangerous to Life and Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ Clayton, G. D.; Clayton, F. E. (編). Patty's Industrial Hygiene and Toxicology. 2A, 2B, 2C: Toxicology 3rd. New York: John Wiley Sons. 1981–1982: 2197.
- ^ Wróblewska, Agnieszka; Drewnowska, E.; Gawarecka, A. The epoxidation of diallyl ether to allyl-glycidyl ether over the TS-1 catalyst. Reaction Kinetics, Mechanisms and Catalysis. August 2016, 118 (2): 719–931. S2CID 101802831. doi:10.1007/s11144-016-1028-3.
| ||||||||||
| 這是一篇與化學相關的小作品。您可以透過編輯或修訂擴充其內容。 |